Adam M Gwozdz, Stephen Black, Beverley J Hunt, Chung S Lim
|
Abstract Venous disease is common in the general population, with chronic venous disorders affecting 50–85% of the western population and consuming 2–3% of healthcare funding. It, therefore, represents a significant socioeconomic, physical and psychological burden. Acute deep vein thrombosis, although a well-recognised cause of death through pulmonary embolism, can more commonly lead to post-thrombotic syndrome (PTS). This article summarises the pathophysiology and risk factor profile of PTS, and highlights various strategies that may reduce the risk of PTS, and the endovenous management of iliofemoral deep vein thrombosis. The authors summarise the advances in PTS risk reduction strategies and present the latest evidence for discussion. Keywords: Post-thrombotic syndrome, deep vein thrombosis, prevention, risk factor, anticoagulant, catheterisation, thrombolytic therapy Disclosure: SAB reports consulting and speakers fees for Cook, Bard, Gore, Veniti, Phillips-Volcano, Medtronic, Boston Scientific and Optimed, and is editor-in-chief of Vascular & Endovascular Review; this did not influence peer review. All other authors have no conflicts of interest to declare. Received: Accepted: Published online: Correspondence Details: Adam M Gwozdz, Academic Department of Vascular Surgery, Guy’s and St Thomas’ NHS Foundation Trust, Westminster Bridge Rd, London SE1 7EH, UK. E: adam.gwozdz@kcl.ac.uk Open Access: This work is open access under the CC-BY-NC 4.0 License which allows users to copy, redistribute and make derivative works for non-commercial purposes, provided the original work is cited correctly. |
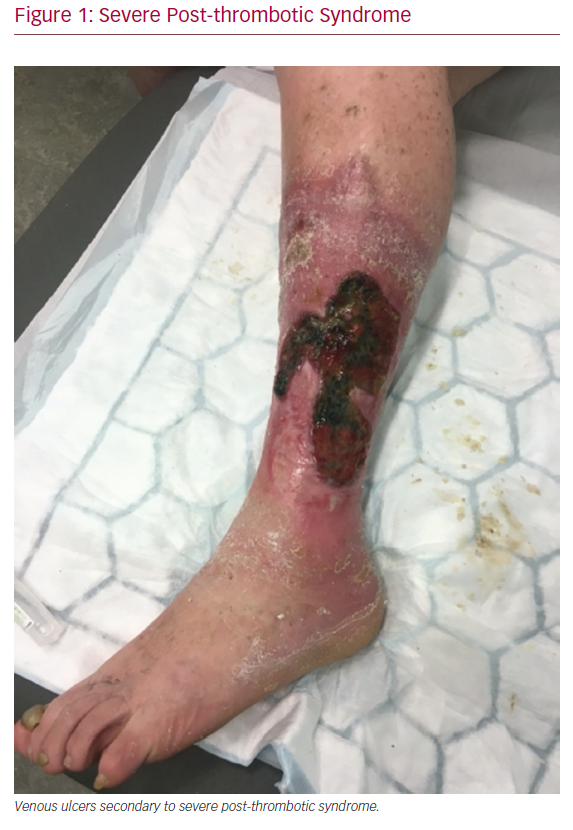
Deep vein thrombosis (DVT) is a common condition estimated to affect approximately 100,000 patients each year in the UK.1 DVT is a well-recognised cause of death through pulmonary embolism (PE), and, rarely, limb loss through phlegmasia cerulea dolens. Most commonly, however, DVT can lead to post-thrombotic syndrome (PTS), which affects patients of all ages, and is characterised by leg pain, itchiness, heaviness, swelling, skin discolouration, and, in severe cases, venous ulceration (Figure 1).2 Severe PTS has major socioeconomic consequences, and even mild PTS can have adverse effects on quality of life (QOL).3,4
Traditionally, anti-coagulation alone was used to prevent the propagation of DVT and PE and allow natural thrombus resolution. However, PTS is increasingly recognised as an important and common debilitating long-term sequela of DVT, given that failure of natural thrombus resolution can lead to a chronically occlusive post-thrombotic limb. PTS can occur in up to approximately 50% of patients in the 2 years after DVT, and is resistant to conservative and early thrombus removal therapies.5,6 Therefore, every effort should be made to reduce the risk of PTS when managing patients with DVT. The aim of this review is to summarise the pathophysiology and risk factors of PTS, highlight various risk reducing strategies for the development of PTS, and discuss future perspectives.
Diagnosis of Post-thrombotic Syndrome
PTS is the most common long-term complication after DVT. PTS is primarily a clinical diagnosis based on the presence of typical symptoms and signs of chronic venous hypertension in a patient with a previous DVT, but no objective diagnostic test exists.7 A number of diagnostic and severity scales have been developed for PTS: the Villalta scale, the Ginsberg measure, the Brandjes scale, the Widmer classification, the Clinical–Etiological–Anatomical–Pathological (CEAP) classification and the Venous Clinical Severity Score (VCSS).8 However, the Villalta scale has been validated externally and endorsed by scientific societies.9 On the Villalta scale, PTS is defined as a score ≥5, or a venous ulcer present, in a leg with previous DVT.10 The Villalta score classifies patients as having or not having PTS, and rates its severity, based on the sum of five venous symptoms and six clinical signs. Mild refers to a Villalta score of 5–9, moderate if the score is 10–14, and severe when the score is ≥15, or if a venous ulcer is present, regardless of the Villalta scoring parameters.10
Pathogenesis of Post-thrombotic Syndrome
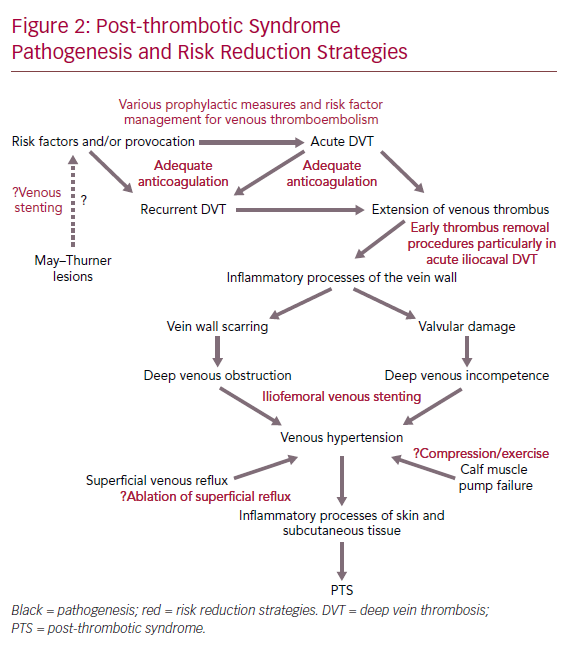
DVT can cause venous outflow obstruction and persistent reflux secondary to vein wall and valvular damage, leading to venous pooling, with limited reversibility depending on the location and extent of thrombus.11–14 This causes changes in microvasculature of the leg, leading to reduced perfusion of surrounding muscle, chronic inflammation, increased vascular permeability and scarring of the vessel wall.7 The presence of venous pooling indirectly affects distal deep veins and superficial collaterals causing dilation and incompetence. As a result, the calf muscle pump becomes ineffective and the ambulatory venous pressure fails to fall significantly with walking or exercise (as it does in the healthy state), which eventually leads to venous hypertension.15 Venous hypertension is thought to initiate chronic inflammatory cascades, which lead to features of PTS including venous claudication, ankle swelling, skin changes and even ulceration.16 However, PTS symptomatology varies over time.3 Figure 2 summarises the pathogenesis of PTS and potential risk reduction strategies.
Severe Post-thrombotic Syndrome
Risk Factors for Post-thrombotic Syndrome
In clinical practice, it would be very useful to be able to predict the individual patient risk of developing PTS and its severity. As a result, prediction tools in the acute and sub-acute phase of DVT are being developed using baseline clinical and demographic characteristics.17–19 These baseline variables include age, BMI, sex, varicose veins, history of venous thrombosis, smoking status, provoked thrombosis and thrombus location. However, further validation is required before these risk scores are used in clinical practice. There are several key factors that increase the risk of developing PTS following DVT (Table 1).7,20–23 Identification of these risk factors, particularly modifiable ones, is important in strategic planning to minimise the risk of developing PTS; preventing DVT from occurring remains the most effective strategy.21 Therefore, it is important that individuals with increased risk of DVT are managed appropriately with the various prophylactic strategies available, such as anti-coagulation, compression hosiery, adequate mobilisation and lifestyle modifications. Strategies for the prevention of DVT are widely available in the literature, hence will not be covered in more detail in this review. Here, we focus on preventative and risk reduction strategies of PTS following DVT occurrence.
Preventative and Risk Reduction Strategies for Post-thrombotic Syndrome
Lifestyle Modification Strategies and Compression
Lifestyle Modifications
There are no studies to support any specific lifestyle modification that may prevent or reduce the risk of PTS. However, in patients with moderate or severe pain initially, early ambulation compared with bed rest was related to remission of acute pain in the affected limb.24 Regular exercise training in patients with PTS was also demonstrated to reduce severity of PTS symptoms and signs.25 Other lifestyle modifications that are likely to improve calf muscle pump, such as weight loss if obese, and frequent leg elevation, relieve some of the symptoms and signs of PTS, and improve wellbeing following DVT. Therefore, all patients with DVT should be counselled on these lifestyle modifications.
Compression
The graduated elastic compression stocking (GECS) has been central to PTS prevention for several decades and is thought to reduce both valvular reflux and venous hypertension.26–29 Although there have been several randomised controlled trials (RCTs) assessing the role of GECS in preventing PTS following DVT, all the studies were limited by their heterogeneity, including the interval between DVT diagnosis and compression, application, type, pressure, duration, co-intervention (in particular the type and duration of anticoagulation), first versus recurrent DVT, PTS diagnostic criteria, and length of follow-up.11,28–34 This heterogeneity complicates comparative analysis, but until recently, the overall evidence seemed to support GECS use for at least 2 years after DVT diagnosis to prevent future PTS.
Risk Factors and Risk Reduction Strategies

This long-held belief in GECS has been challenged by evidence from the Compression Stockings to Prevent the Post-Thrombotic Syndrome (SOX) trial; a placebo-controlled, multicentre RCT that showed no benefit of GECS in preventing PTS.34 In the RCT, cumulative incidence of PTS was 14.2% with GECS compared with 12.7% in the placebo group. However, the trial was criticised for the low GECS compliance, with only 55.6% of patients wearing the GECS for ≥3 days per week at 2 years.
Two other studies with high GECS compliance demonstrated decreased PTS incidence, although it is unclear whether these studies included iliofemoral DVTs.30,31 This may suggest that GECS worn in a manner reflective of patient daily practice does not prevent PTS.35 As a result, there are some variations in the recommendation of the use of GECS following DVT among international and national guidelines. Overall, GECS may be used for symptom relief but the evidence of its role in preventing PTS is uncertain.36 In 2012, the National Institute for Health and Care Excellence (NICE) recommended offering below-knee GECS with ankle pressure >23 mmHg ≤3 weeks after the diagnosis of iliofemoral DVT. However, in 2015, following review of the SOX trial, NICE updated its guideline, and advised not to offer GECS following iliofemoral DVT for the prevention of PTS, but to use GECS only for symptomatic relief.37
More recently, the One versus Two Years of Elastic Compression Stockings for Prevention of Post-thrombotic Syndrome (OCTAVIA) study showed that stopping GECS after 1 year in patients with proximal DVT seemed to be non-inferior to continuing GECS for 2 years.38 In 2018, the Individualised versus Standard Duration of Elastic Compression Therapy for Prevention of Post-thrombotic Syndrome (IDEAL DVT) non-inferiority study showed that it was safe to shorten the duration of GECS on an individualised basis after proximal DVT for prevention of PTS.39 A further RCT, the multicentre Compression Hosiery to Avoid Post-thrombotic Syndrome (CHAPS) study (ISRCTN73041168) in the UK aims to address the effectiveness of GECS in preventing PTS in patients with DVT.40
Intermittent pneumatic compression (IPC) devices apply variable pressure cycles on the lower limb with inflatable compartments to emulate the calf muscle pump. Physiologically, IPC is thought to protect against venous thromboembolism (VTE) in a variety of ways: by reducing venous stasis, inducing flow-related venous endothelial alterations, improving lymphatic drainage and increasing endogenous fibrinolytic potential. A series of RCTs and meta-analyses have shown that IPC use alone reduces DVT incidence by more than 60%, with further reduction when concurrent pharmacological prophylaxis is used.41 These data have been supported by a 2016 Cochrane review that confirmed that, based on moderate quality evidence, IPC plus pharmacological prophylactic measures decreased PE incidence when compared with anticoagulation alone and decreased the incidence of DVT compared with GECS alone.42 Therefore, the usage of IPC to prevent postoperative DVT, particularly in high-risk cases, should be considered as part of a multi-modal PTS prevention strategy.
Medical Strategies
Anticoagulation
The use of anticoagulation after first acute DVT has the largest, proven benefit in reducing the incidence of PTS when compared with no treatment. Systemic anticoagulation therapy following DVT prevents the propagation of existing thrombi, formation of new DVT, PE, and recurrent DVT, all of which are known risk factors for the development of PTS.43 However, anticoagulation cannot lyse acute thrombus; this depends on the patient’s endogenous fibrinolytic system.44
Current practice following acute DVT is use of low-molecular-weight heparin followed by bridging to dose-adjusted oral vitamin K antagonists (VKA), such as warfarin, until an international normalised ratio (INR) target of 2–3 is achieved, at which point warfarin only is continued; or the use of a direct oral anticoagulant (DOAC) from day 1 with or without bridging parenteral therapy.45 The time in therapeutic range is critical to the effectiveness; for example, in warfarin therapy monitored using INR, a subtherapeutic anticoagulation (defined as INR <2 for >20% of the time) was associated with a significant increase in PTS development.46
Recently it was found that treatment of DVT with rivaroxaban might be associated with a lower risk for PTS development.47–49 In 2020, in a study of patients with acute proximal DVT, the risk of PTS in the DOAC-treated patients was reduced by 54% compared with patients treated with VKA (OR 0.46; 95% CI [0.33–0.63]).50 Although the authors advise interpreting the results with caution, they propose that patients treated with a DOAC, unlike those receiving VKAs, have progressively increased vein recanalisation over time. Overall, the anticoagulation strategy should be tailored to the individual, taking into account patient preference and compliance, comorbidities, polypharmacy, bleeding risk, DVT aetiology and risk of recurrence.51
Other Drugs
There is limited evidence to support venoactive drugs, such as rutosides (a herbal remedy used in chronic venous insufficiency to reduce swelling and skin changes), defibrotide (a single-stranded polydeoxyribonucleotide that has anti-thrombotic, anti-inflammatory and anti-ischaemic properties) and hidrosmin (a vasoprotective synthetic bioflavonoid) in preventing PTS following DVT, hence their use is not recommended at present.52,53 Sulodexide, a glycosaminoglycan consisting of unbranched polysaccharide chains with numerous biological effects including anti-thrombogenesis, anti-inflammatory effects, and endothelial protection, has recently been shown to potentially reduce recurrent VTE and PTS, although further clinical trials are needed to confirm its roles.54,55
Stategies for Early Thrombus Removal
There are two main indications for early removal of thrombus in patients with acute DVT. First, removal of thrombus may be needed in patients with severe pain and swelling, especially when there is increased risk of limb-threatening ischaemia such as phlegmasia cerulea dolens, or worsening symptoms despite optimal medical and conservative treatment. Second, early removal of thrombus in patients with iliofemoral DVT may reduce the risk of PTS development, as a result of the reduction in inflammation and injury to the vein wall and valves that would otherwise be caused by residual thrombus.
Several studies, including RCTs, reported that in selected patients, removal of acute thrombus resulted in better long-term outcomes compared with conservative measures alone in terms of reducing the risk of PTS.56–59 However, two recent multicentre RCTs have questioned the efficacy of early removal of thrombus to reduce PTS, although both studies were limited by various methodological flaws.6,60 Major guidance from NICE, and the American Venous Forum/Society for Vascular Surgery (AVF/SVS) recommend consideration of thrombus removal intervention in the 14 days after acute DVT; tissue plasminogen activator (tPA) has the greatest effect in experimental animal thrombi when fibrin content is greatest, between 7 and 10 days following induction.61
Surgical Thrombectomy
Until the 1970s, the principal method of removal of thrombus in acute DVT was surgical thrombectomy.62 Surgical thrombectomy did not show long-term benefits until it was combined with arteriovenous fistula formation distal to the site of venous reconstruction in order to improve venous inflow.63,64 However, this was suitable only for a select group of patients, and thrombosis recurred early if residual thrombus remained after the procedure. Surgical thrombectomy is not routinely performed, largely due to the invasiveness of the procedure, and to the significant potential for morbidity compared with percutaneous interventions. However, occasionally it is still indicated, particularly in patients with acute DVT requiring rapid removal of thrombus to relieve a limb-threatening ischaemia.
Systemic Thrombolysis
Systemic thrombolysis demonstrates superior clot lysis in acute DVT patients compared with conservative treatment alone.65 However, systemic thrombolysis (dose used varied; streptokinase the most common agent used, with and without heparin) was associated with a high rate of major bleeding complications such as intracranial haemorrhage and retroperitoneal haematoma.65 As such, systemic thrombolysis is not recommended in current practice.66 Furthermore, an early observation from Meyerovitz et al. showed that systemic treatment with thrombolytic agent did not permit sufficient penetration into occluded thrombi, a challenge that catheter-directed thrombolysis (CDT) and pharmacomechanical thrombolysis (PMT) were developed to overcome.67
Catheter-directed Thrombolysis
CDT was developed as a minimally invasive procedure with the aim of removing the bulk of the thrombus, leaving an ‘open vein’ with no obstruction to venous flow.68,69 In doing so, CDT has overcome many of the limitations of a systemic agent.70 The procedure involves ultrasound identification of a suitable vein for access (typically either popliteal, femoral or, more rarely, internal jugular vein) followed by introduction of a catheter into the deep venous system. This allows for targeted delivery of high concentrations of a fibrinolytic agent, such as tPA, directly into the occlusion site via a multi-sidehole catheter.
In our practice, a 10 mg bolus of alteplase is infused throughout the thrombus followed by 1 mg/h for 5 hours. A check venogram will generally be carried out after 12–24 h to assess the degree of thrombus dissolution, and evaluate the need for repeat thrombolysis and adjunctive angioplasty or stenting (Figure 3).71,72 When considering CDT, patient preference, as well as bleeding risk and comorbidities must be taken into account. Location and extent of DVT is important, given that isolated calf DVT has a much lower risk of PTS compared with iliocaval extension.28,73
Early studies demonstrated a reduction in incidence of PTS following CDT.56,74,75 The Catheter-directed Venous Thrombolysis in Acute Iliofemoral Vein Thrombosis (CaVenT) study, a multicentre RCT of 209 patients comparing CDT with standard treatment alone (anti-coagulation and compression), found significantly improved iliofemoral patency rates (65.9%) in patients treated with CDT (65.9% versus 47.4%, p=0.012).76 Additionally, after 2 and 5 years, there was a significant absolute risk reduction of 14.4% (41.1% in CDT versus 55.6% in control, 95% CI [0.2–27.9]; number needed to treat of 7, 95% CI [4–502]), and 28% (43% in CDT versus 71% in control, 95% CI [14–42]; number needed to treat of 4, 95% CI [2–7]), respectively. Interestingly, QOL at 5 years as measured with the EuroQol-5 Dimension (EQ-5D) and the disease-specific VEnous INsufficiency Epidemiological and Economic Study (VEINES)–Quality of Life/Symptoms (VEINES-QOL/Sym) questionnaires did not differ between treatment groups.77
CDT is recommended in the NICE and AVF/SVS guidelines for treatment of symptomatic iliofemoral DVT <14 days old, in patients with good functional capacity, life expectancy of more than 1 year and low risk of bleeding. However, potential limitations of CDT include time delay to lysis and hospital stay in a high dependency unit, with the associated economic implications, although this needs further research.66,78–80
Mechanical and Aspiration Thrombectomy
Mechanical and aspiration thrombectomy provides an alternate minimally invasive method of thrombus removal in the case of contraindications to pharmacological thrombolysis. Potential contraindications to pharmacological thrombolysis, such as CDT, include recent major surgery, trauma, stroke, pregnancy and active or recent bleeding. As the name implies, this form of thrombectomy uses mechanical force generated through rotatory and rheolytic means to break up the thrombus into smaller segments that can often then be aspirated. For example, a modern derivative of a Fogarty balloon is inserted distal to the thrombus and then retrieved with the thrombus.81,82 The rate of PE following surgical thrombectomy is <1%, which is comparable to the incidence in conservatively treated patients.83
Endovascular Therapy for Acute Iliac Vein Thrombosis
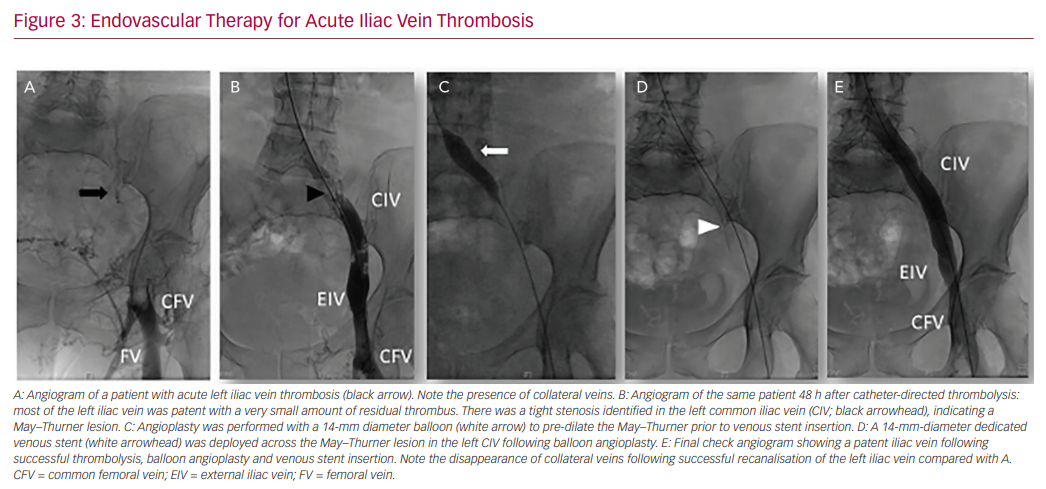
Several types of pharmacomechanical catheter-directed therapy have been developed to improve the efficiency of thrombus clearance compared with CDT or mechanical thrombectomy alone. PMT consists of an endovascular device that is advanced into the thrombus, which performs a combination of maceration and/or aspiration to physically break down the thrombus. This increases the surface area of residual thrombus for both exogenous and endogenous thrombolytic processes, reducing both the dose of thrombolytic agent required and the duration of thrombolysis.84–86 Limited evidence thus far suggests PMT is safe and effective in reducing PTS.87
The Acute Venous Thrombosis: Thrombus Removal with Adjunctive Catheter-directed Thrombolysis (ATTRACT) trial set out to address the treatment effect of adding PMT (catheter mediated or device mediated, with or without venous stenting) compared with anticoagulation alone on the incidence of PTS, measured with the Villalta score, in patients with acute proximal DVT.6 In all, 692 patients with acute proximal DVT were randomised to receive either anticoagulation alone or anticoagulation plus PMT. The ATTRACT trial reported no significant between-group difference in the percentage of patients with PTS between 6 and 24 months (48% versus 47%, respectively, p=0.56). Importantly, a significant reduction in moderate-to-severe PTS in patients receiving early thrombus removal therapy was observed (18% in the PMT group versus 24% of those in the anticoagulation alone group, p=0.04). Furthermore, when PTS was continuously assessed at 6, 12, 18 and 24 months after treatment, symptom severity scores at each follow-up were significantly lower in patients who had received PMT compared with anticoagulation alone (p<0.01).6 This suggests that although PMT did not prevent the onset of PTS, it resulted in significant symptom improvement. However, optimal timing of the intervention from the onset of DVT remains unclear.
A further post-hoc analysis of ATTRACT patients with iliofemoral DVT examined the effect of PMT.88 Although PMT did not influence the occurrence of PTS, it significantly reduced early leg symptoms, and, over 24 months, reduced PTS severity scores, and the proportion of patients who developed moderate-or-severe PTS. However, a limitation of this analysis was the substantial loss to follow-up that was unbalanced between the treatment groups (more missed PTS assessments in the non-PMT arm), which influenced the study’s estimates of treatment effects.6 More recently, the Dutch Catheter Versus Anticoagulation Alone for Acute Primary (Ilio)Femoral DVT (CAVA) trial with 184 participants randomised to ultrasound-accelerated CDT versus standard care only (anticoagulation, knee-high ECS and early ambulation) reported no significant difference in 1 year rates of PTS between the two groups (29% versus 35%).60 However, that study carried several limitations including a relatively small proportion of iliofemoral DVT patients and a low procedural technical success rate.
In the AVF/SVS guidelines, early thrombus removal with PMT over CDT is recommended ≤14 days after acute iliofemoral DVT if resources and expertise allow, due to improved efficacy and the more favourable safety profile.66 According to recent NICE recommendations, percutaneous mechanical thrombectomy for acute DVT of the leg has well-recognised but infrequent complications; hence the procedure should be used only with special arrangements for clinical governance, consent, and audit or research.89
Adjuncts to Thrombus Removal Procedures
Endovenous Balloon Angioplasty and Deep Venous Stenting
This is a growth area as technologies are being developed to remove ‘old clot’ from patients with established PTS. Old clot, however, contains little residual thrombus but is replaced by fibrous tissue.90 Endovenous balloon angioplasty and stenting of deep venous (particularly common femoral vein, iliac vein, and inferior vena cava) residual obstructive lesions after early thrombus removal procedures such as CDT and PMT are increasingly favoured.65,90 Venogram and/or intravascular ultrasound (IVUS) is used to identify and measure the degree and extent of obstructive lesions prior to balloon angioplasty and stent insertion. Although endovenous balloon angioplasty and stenting may reduce the risk of recurrent DVT and PTS in some patients, there is as yet no high-quality evidence to support the routine use of such procedures. Further research is required to identify the group of patients who may benefit from such adjunct intervention, and the optimal time and degree of venous stenosis for endovenous balloon angioplasty and stenting, and its cost effectiveness.
Due to the nature of the obstructive fibrotic and compressive lesions of the vein wall, balloon angioplasty alone is not sufficient, hence insertion of self-expandable stent is required to maintain the intended lumen diameter. Until recently, only stents designed for arterial pathology were used. However, several dedicated venous stents are currently available. These dedicated venous stents are made from nitinol (a metal alloy of nickel and titanium), with a size (diameter and length), strength, flexibility, and resistance to thrombosis tailored to the venous system and pathology, which differ from their arterial counterparts.91 Patency rates of dedicated venous stents at 12 and 24 months are encouraging,92,93 but longer-term results are awaited.
The abovementioned ATTRACT study, and CAVA trial both include venoplasty and/or stenting at the discretion of the operator following PMT in their management protocols.60 In a small prospective study looking specifically at venous stenting after CDT in extrinsic compression of iliac vein (e.g. May–Thurner or Cockett’s syndrome), the acute phase patency rate was 92.3%, and the mid-term patency rate was 90%.94 These non-occlusive, non-thrombotic lesions are significantly easier to treat compared with their thrombotic occlusive counterparts. May–Thurner or Cockett’s syndrome is most often characterised by extrinsic compression of the left common iliac vein by the overlying right common iliac artery, but compression may occur at multiple sites and commonly affects the left lower limb.
Future Research and Perspectives
Despite PTS being common and causing much physical, social and work-related morbidity, there has been little research interest in this area until recently. More research is required to understand, prevent and manage established PTS. Improving our understanding of the pathogenesis and natural history of DVT and PTS, including at the molecular level, will help in the identification and modification of the risk factors involved in DVT and PTS. Various potential molecular markers including d-dimer, factor VIII, soluble thrombomodulin, tPA and specific genetic and inflammatory markers are currently being investigated for their prognostic value in PTS.22,23,95,96 Developing objective and validated risk stratifications may help identify high-risk patients who may benefit from more aggressive measures to reduce the risk of PTS. Early removal of thrombus is associated with reduction of PTS risk in patients with acute iliofemoral DVT through effective recanalisation of the venous system, and reduction of venous wall and valve injury. There is also evidence from RCTs to support the role of early CDT in reducing the rate of PTS in patients with acute iliofemoral DVT. Further consensus and guidance are also needed in postoperative anticoagulation strategies to maintain long-term stent patency. Advances in imaging technology have provided opportunities to develop modalities that are able to characterise the thrombus.
Saha et al. are currently researching the use of magnetic resonance in direct thrombus imaging in measuring the age of the thrombus, which may help with patient selection for endovenous therapies.61 Simple measures, including compression and regular exercise, still require further high-quality trials to clarify their roles in reducing the risk of PTS development. Advancements in endovascular technology, such as PMT and mechanical thrombectomy devices, and dedicated venous stents, have provided enormous research opportunities into the prevention and improved management of established PTS. Research on bioprosthetic venous valves is also potentially helpful in the prevention of PTS.
- Cohen AT, Agnelli G, Anderson FA, et al. Venous thromboembolism in Europe. The number of VTE events and associated morbidity and mortality. Thromb Haemost 2007;98:756–64.
Crossref| PubMed - Eklof B, Perrin M, Delis KT, et al. Updated terminology of chronic venous disorders: the VEIN-TERM transatlantic interdisciplinary consensus document. J Vasc Surg 2009;49:498–501.
Crossref| PubMed - Kahn SR, Shrier I, Julian JA, et al. Determinants and time course of the postthrombotic syndrome after acute deep venous thrombosis. Ann Intern Med 2008;149:698–707.
Crossref| PubMed - Vedantham S, Kahn SR, Goldhaber SZ, et al. Endovascular therapy for advanced post-thrombotic syndrome: proceedings from a multidisciplinary consensus panel. Vasc Med 2016;21:400–407.
Crossref| PubMed - Cohen A, Agnelli G, Anderson F, et al. Venous thromboembolism (VTE) in Europe. Thromb Haemost 2007;98:756–64.
Crossref| PubMed - Vedantham S, Goldhaber SZ, Julian JA, et al. Pharmacomechanical catheter-directed thrombolysis for deep-vein thrombosis. N Engl J Med 2017;377:2240–52.
Crossref| PubMed - Kahn SR. The post-thrombotic syndrome. Hematology Am Soc Hematol Educ Program 2016;2016:413–18.
Crossref| PubMed - Wik HS, Enden TR, Ghanima W, et al. Diagnostic scales for the post-thrombotic syndrome. Thromb Res 2018;164:110–15.
Crossref| PubMed - Lattimer CR, Kalodiki E, Azzam M, Geroulakos G. Validation of the Villalta scale in assessing post-thrombotic syndrome using clinical, duplex, and hemodynamic comparators. J Vasc Surg Venous Lymphat Disord 2014;2:8–14.
Crossref| PubMed - Kahn SR, Partsch H, Vedantham S, et al. Definition of post-thrombotic syndrome of the leg for use in clinical investigations: a recommendation for standardization. J Thromb Haemost 2009;7:879–83.
Crossref| PubMed - Roumen-Klappe EM, Janssen MCH, Van Rossum J, et al. Inflammation in deep vein thrombosis and the development of post-thrombotic syndrome: a prospective study. J Thromb Haemost 2009;7:582–7.
Crossref| PubMed - Markel A, Manzo RA, Bergelin RO, Strandness DE Jr. Valvular reflux after deep vein thrombosis: incidence and time of occurrence. J Vasc Surg 1992;15:377–84.
Crossref| PubMed - Caps MT, Manzo RA, Bergelin RO, et al. Venous valvular reflux in veins not involved at the time of acute deep vein thrombosis. J Vasc Surg 1995;22:524–31.
Crossref| PubMed - Prandoni P, Frulla M, Sartor D, et al. Vein abnormalities and the post-thrombotic syndrome. J Thromb Haemost 2005;3:401–2.
Crossref| PubMed - Christopoulos D, Nicolaides AN, Cook A, et al. Pathogenesis of venous ulceration in relation to the calf muscle pump function. Surgery 1989;106:829–35.
PubMed - Busuttil A, Lim CS, Davies AH. Post thrombotic syndrome. Adv Exp Med Biol 2017;906:363–75.
Crossref| PubMed - Méan M, Limacher A, Alatri A, et al. Derivation and validation of a prediction model for risk stratification of post-thrombotic syndrome in elderly patients with a first deep vein thrombosis. Thromb Haemost 2018;118:1419–27.
Crossref| PubMed - Amin EE, van Kuijk SMJ, Joore MA, et al. Development and validation of a practical two-step prediction model and clinical risk score for post-thrombotic syndrome. Thromb Haemost 2018;118:1242–9.
Crossref| PubMed - Rabinovich A, Ducruet T, Kahn SR, SOX Trial investigators. Development of a clinical prediction model for the postthrombotic syndrome in a prospective cohort of patients with proximal deep vein thrombosis. J Thromb Haemost 2017;16:262–70.
Crossref| PubMed - Galanaud J-P, Monreal M, Kahn SR. Predictors of the post-thrombotic syndrome and their effect on the therapeutic management of deep vein thrombosis. J Vasc Surg Venous Lymphat Disord 2016;4:531–4.
Crossref| PubMed - Kahn SR, Galanaud J-P, Vedantham S, Ginsberg JS. Guidance for the prevention and treatment of the post-thrombotic syndrome. J Thromb Thrombolysis 2016;41:144–53.
Crossref| PubMed - Sartori M, Favaretto E, Cini M, et al. D-dimer, FVIII and thrombotic burden in the acute phase of deep vein thrombosis in relation to the risk of post-thrombotic syndrome. Thromb Res 2014;134:320–5.
Crossref| PubMed - Bouman AC, Cheung YW, Spronk HM, et al. Biomarkers for post thrombotic syndrome: a case-control study. Thromb Res 2014;134:369–75.
Crossref| PubMed - Liu Z, Tao X, Chen Y, et al. Bed rest versus early ambulation with standard anticoagulation in the management of deep vein thrombosis: a meta-analysis. PLoS One 2015;10:e0121388.
Crossref| PubMed - Kahn SR, Shrier I, Shapiro S, et al. Six-month exercise training program to treat post-thrombotic syndrome: a randomized controlled two-centre trial. CMAJ 2011;183:37–44.
Crossref| PubMed - Pierson S, Pierson D, Swallow R, et al. Efficacy of graded elastic compression in the lower leg. JAMA 1983;249:242–3.
Crossref| PubMed - Brakkee AJM, Kuiper JP. The influence of compressive stockings on the haemodynamics in the lower extremities. Phlebology 1988;3:147–54.
Crossref - Ginsberg JS, Hirsh J, Julian J, et al. Prevention and treatment of postphlebitic syndrome: results of a 3-part study. Arch Intern Med 2001;161:2105–9.
Crossref| PubMed - Partsch H, Kaulich M, Mayer W. Immediate mobilisation in acute vein thrombosis reduces post-thrombotic syndrome. Int Angiol 2004;23:206–12.
PubMed - Brandjes DP, Büller HR, Heijboer H, et al. Randomised trial of effect of compression stockings in patients with symptomatic proximal-vein thrombosis. Lancet 1997;349:759–62.
Crossref| PubMed - Prandoni P, Lensing AWA, Prins MH, et al. Below-knee elastic compression stockings to prevent the post-thrombotic syndrome: a randomized, controlled trial. Ann Intern Med 2004;141:249–56.
Crossref| PubMed - Aschwanden M, Jeanneret C, Koller MT, et al. Effect of prolonged treatment with compression stockings to prevent post-thrombotic sequelae: a randomized controlled trial. J Vasc Surg 2008;47:1015–21.
Crossref| PubMed - Jayaraj A, Meissner M. Impact of graduated compression stockings on the prevention of post-thrombotic syndrome: results of a randomized controlled trial. Phlebology 2015;30:541–8.
Crossref| PubMed - Kahn SR, Shapiro S, Wells PS, et al. Compression stockings to prevent post-thrombotic syndrome: a randomised placebo-controlled trial. Lancet 2014;383:880–8.
Crossref| PubMed - Raju S, Hollis K, Neglén P. Use of compression stockings in chronic venous disease: patient compliance and efficacy. Ann Vasc Surg 2007;21:790–5.
Crossref| PubMed - Righini M, Robert-Ebadi H, Glauser F, et al. Effect of anticoagulant treatment on pain in distal deep vein thrombosis: an ancillary analysis from the cactus trial. J Thromb Haemost 2019;17:507–10.
Crossref| PubMed - National Institute for Health and Care Excellence. Venous thromboembolic diseases: diagnosis, management and thrombophilia testing (update). CG144. London: NICE, 2015.
- Mol GC, van de Ree MA, Klok FA, et al. One versus two years of elastic compression stockings for prevention of post-thrombotic syndrome (OCTAVIA study): randomised controlled trial. BMJ 2016;353:i2691.
Crossref| PubMed - Cate-Hoek ten AJ, Amin EE, Bouman AC, et al. Individualised versus standard duration of elastic compression therapy for prevention of post-thrombotic syndrome (IDEAL DVT): a multicentre, randomised, single-blind, allocation-concealed, non-inferiority trial. Lancet Haematol 2018;5:e25–33.
Crossref| PubMed - ISRCTN registry. Graduated compression stockings for patients with a deep vein thrombosis to prevent long-term symptoms, known as post-thrombotic syndrome [ISRCTN73041168]. 2019.
Crossref - Delos Reyes AP, Partsch H, Mosti G, et al. Report from the 2013 meeting of the International Compression Club on advances and challenges of compression therapy. J Vasc Surg Venous Lymphat Disord 2014;2:469–76.
Crossref| PubMed - Kakkos SK, Caprini JA, Geroulakos G, et al. Combined intermittent pneumatic leg compression and pharmacological prophylaxis for prevention of venous thromboembolism. Cochrane Database Syst Rev 2016;(4):CD005258.
Crossref| PubMed - Kearon C, Kahn SR, Agnelli G, et al. Antithrombotic therapy for venous thromboembolic disease: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines (8th Edition). Chest 2008;133(6 Suppl):454S–545S.
Crossref| PubMed - Comerota AJ, Gravett MH. Iliofemoral venous thrombosis. J Vasc Surg 2007;46:1065–76.
Crossref| PubMed - Black SA, Cohen AT. Anticoagulation strategies for venous thromboembolism: moving towards a personalised approach. Thromb Haemost 2015;114:660–9.
Crossref| PubMed - Chitsike RS, Rodger MA, Kovacs MJ, et al. Risk of post-thrombotic syndrome after subtherapeutic warfarin anticoagulation for a first unprovoked deep vein thrombosis: results from the REVERSE study. J Thromb Haemost 2012;10:2039–44.
Crossref| PubMed - Jeraj L, Jezovnik MK, Poredos P. Rivaroxaban versus warfarin in the prevention of post-thrombotic syndrome. Thromb Res 2017;157:46–8.
Crossref| PubMed - Søgaard M, Nielsen PB, Skjøth F, et al. Rivaroxaban versus warfarin and risk of post-thrombotic syndrome among patients with venous thromboembolism. Am J Med 2018;131:787–94.e4.
Crossref| PubMed - Utne KK, Dahm A, Wik HS, et al. Rivaroxaban versus warfarin for the prevention of post-thrombotic syndrome. Thromb Res 2018;163:6–11.
Crossref| PubMed - Prandoni P, Ageno W, Ciammaichella M, et al. The risk of post-thrombotic syndrome in patients with proximal deep vein thrombosis treated with the direct oral anticoagulants. Intern Emerg Med 2020;15:447–52.
Crossref| PubMed - Konstantinides SV. 2014 ESC Guidelines on the diagnosis and management of acute pulmonary embolism. Eur Heart J 2014;35:3145–6.
Crossref| PubMed - Morling JR, Yeoh SE, Kolbach DN. Rutosides for treatment of post-thrombotic syndrome. Cochrane Database Syst Rev 2015;(9):CD005625.
Crossref| PubMed - Cohen JM, Akl EA, Kahn SR. Pharmacologic and compression therapies for postthrombotic syndrome: a systematic review of randomized controlled trials. Chest 2012;141:308–20.
Crossref| PubMed - Jiang Q-J, Bai J, Jin J, et al. Sulodexide for secondary prevention of recurrent venous thromboembolism: a systematic review and meta-analysis. Front Pharmacol 2018;9:876.
Crossref| PubMed - Luzzi R, Belcaro G, Dugall M, et al. The efficacy of sulodexide in the prevention of postthrombotic syndrome. Clin Appl Thromb Hemost 2014;20:594–9.
Crossref| PubMed - Elsharawy M, Elzayat E. Early results of thrombolysis vs anticoagulation in iliofemoral venous thrombosis. A randomised clinical trial. Eur J Vasc Endovasc Surg 2002;24:209–14.
Crossref| PubMed - Sharifi M, Bay C, Mehdipour M, Sharifi J for the TORPEDO Investigators. Thrombus Obliteration by Rapid Percutaneous Endovenous Intervention in Deep Venous Occlusion (TORPEDO) Trial: midterm results. J Endovasc Ther 2012;19:273–80.
Crossref| PubMed - Enden T, Wik HS, Kvam AK, et al. Health-related quality of life after catheter-directed thrombolysis for deep vein thrombosis: secondary outcomes of the randomised, non-blinded, parallel-group CaVenT study. BMJ Open 2013;3:e002984–7.
Crossref| PubMed - Sharifi M, Mehdipour M, Bay C, et al. Endovenous therapy for deep venous thrombosis: the TORPEDO Trial. Catheter Cardiovasc Interv 2010;76:316–25.
Crossref| PubMed - Notten P, Cate-Hoek ten AJ, Arnoldussen CWKP, et al. Ultrasound-accelerated catheter-directed thrombolysis versus anticoagulation for the prevention of post-thrombotic syndrome (CAVA): a single-blind, multicentre, randomised trial. Lancet Haematol 2020;7:e40–9.
Crossref| PubMed - Saha P, Andia ME, Modarai B, et al. Magnetic resonance T1 relaxation time of venous thrombus is determined by iron processing and predicts susceptibility to lysis. Circulation 2013;128:729–36.
Crossref| PubMed - Deweese JA. Thrombectomy for acute iliofemoral venous thrombosis. J Cardiovasc Surg (Torino) 1964;5:703–12.
PubMed - Menawat SS, Gloviczki P, Mozes G, et al. Effect of a femoral arteriovenous fistula on lower extremity venous hemodynamics after femorocaval reconstruction. J Vasc Surg 1996;24:793–9.
Crossref| PubMed - Juhan CM, Alimi YS, Barthelemy PJ, et al. Late results of iliofemoral venous thrombectomy. J Vasc Surg 1997;25:417–22.
Crossref| PubMed - Schweizer J, Kirch W, Koch R, et al. Short- and long-term results after thrombolytic treatment of deep venous thrombosis. J Am Coll Cardiol 2000;36:1336–43.
Crossref| PubMed - Meissner MH, Gloviczki P, Comerota AJ, et al. Early thrombus removal strategies for acute deep venous thrombosis: clinical practice guidelines of the Society for Vascular Surgery and the American Venous Forum. J Vasc Surg 2012;55:1449–62.
Crossref| PubMed - Meyerovitz MF, Polak JF, Goldhaber SZ. Short-term response to thrombolytic therapy in deep venous thrombosis: predictive value of venographic appearance. Radiology 1992;184:345–8.
Crossref| PubMed - Comerota AJ. Catheter-directed thrombolysis prevents post-thrombotic syndrome in patients with acute deep vein thrombosis in the upper half of the thigh. Evid Based Med 2012;17:182–3.
Crossref| PubMed - Vedantham S. Thrombolytic therapy for deep vein thrombosis. Curr Treat Options Cardiovasc Med 2009;11:129–35.
Crossref| PubMed - Vedantham S. Endovascular procedures in the management of DVT. Hematology Am Soc Hematol Educ Program 2011;2011:156–61.
Crossref| PubMed - Semba CP, Dake MD. Iliofemoral deep venous thrombosis: aggressive therapy with catheter-directed thrombolysis. Radiology 1994;191:487–94.
Crossref| PubMed - Vedantham S, Millward SF, Cardella JF, et al. Society of Interventional Radiology position statement: treatment of acute iliofemoral deep vein thrombosis with use of adjunctive catheter-directed intrathrombus thrombolysis. J Vasc Interv Radiol 2006;17:613–16.
Crossref| PubMed - Vedantham S, Sista AK, Klein SJ, et al. Quality improvement guidelines for the treatment of lower-extremity deep vein thrombosis with use of endovascular thrombus removal. J Vasc Interv Radiol 2014;25:1317–25.
Crossref| PubMed - Comerota AJ, Throm RC, Mathias SD, et al. Catheter-directed thrombolysis for iliofemoral deep venous thrombosis improves health-related quality of life. J Vasc Surg 2000;32:130–7.
Crossref| PubMed - AbuRahma AF, Perkins SE, Wulu JT, Ng HK. Iliofemoral deep vein thrombosis: conventional therapy versus lysis and percutaneous transluminal angioplasty and stenting. Ann Surg 2001;233:752–60.
Crossref| PubMed - Enden T, Haig Y, Kløw N-E, et al. Long-term outcome after additional catheter-directed thrombolysis versus standard treatment for acute iliofemoral deep vein thrombosis (the CaVenT study): a randomised controlled trial. Lancet 2012;379:31–8.
Crossref| PubMed - Haig Y, Enden T, Grøtta O, et al. Post-thrombotic syndrome after catheter-directed thrombolysis for deep vein thrombosis (CaVenT): 5-year follow-up results of an open-label, randomised controlled trial. Lancet Haematol 2016;3:e64–71.
Crossref| PubMed - Nazir SA, Ganeshan A, Nazir S, Uberoi R. Endovascular treatment options in the management of lower limb deep venous thrombosis. Cardiovasc Intervent Radiol 2009;32:861–76.
Crossref| PubMed - Mewissen MW, Seabrook GR, Meissner MH, et al. Catheter-directed thrombolysis for lower extremity deep venous thrombosis: report of a national multicenter registry. Radiology 1999;211:39–49.
Crossref| PubMed - Baekgaard N, Broholm R, Just S, et al. Long-term results using catheter-directed thrombolysis in 103 lower limbs with acute iliofemoral venous thrombosis. Eur J Vasc Endovasc Surg 2010;39:112–17.
Crossref| PubMed - Kwon SH, Oh JH, Seo T-S, et al. Percutaneous aspiration thrombectomy for the treatment of acute lower extremity deep vein thrombosis: is thrombolysis needed? Clin Radiol 2009;64:484–90.
Crossref| PubMed - Oguzkurt L, Ozkan U, Gülcan O, et al. Endovascular treatment of acute and subacute iliofemoral deep venous thrombosis by using manual aspiration thrombectomy: long-term results of 139 patients in a single center. Diagn Interv Radiol 2012;18:410–6.
Crossref| PubMed - Lindow C, Mumme A, Asciutto G, et al. Long-term results after transfemoral venous thrombectomy for iliofemoral deep venous thrombosis. Eur J Vasc Endovasc Surg 2010;40:134–8.
Crossref| PubMed - Kasirajan K, Gray B, Ouriel K. Percutaneous AngioJet thrombectomy in the management of extensive deep venous thrombosis. J Vasc Interv Radiol 2001;12:179–85.
Crossref| PubMed - Lin PH, Ochoa LN, Duffy P. Catheter-directed thrombectomy and thrombolysis for symptomatic lower-extremity deep vein thrombosis: review of current interventional treatment strategies. Perspect Vasc Surg Endovasc Ther 2010;22:152–63.
Crossref| PubMed - Pouncey AL, Gwozdz AM, Johnson OW, et al. AngioJet pharmacomechanical thrombectomy and catheter directed thrombolysis vs. catheter directed thrombolysis alone for the treatment of iliofemoral deep vein thrombosis: a single centre retrospective cohort study. Eur J Vasc Endovasc Surg 2020;60:578–85.
Crossref| PubMed - Karthikesalingam A, Young EL, Hinchliffe RJ, et al. A systematic review of percutaneous mechanical thrombectomy in the treatment of deep venous thrombosis. Eur J Vasc Endovasc Surg 2011;41:554–65.
Crossref| PubMed - Comerota AJ, Kearon C, Gu C-S, et al. Endovascular thrombus removal for acute iliofemoral deep vein thrombosis. Circulation 2019;139:1162–73.
Crossref| PubMed - National Institute for Health and Care Excellence. Percutaneous mechanical thrombectomy for acute deep vein thrombosis of the leg. IPG651. London: NICE, 2019. https://www.nice.org.uk/guidance/ipg651 (accessed 6 December 2020).
- Comerota AJ, Oostra C, Fayad Z, et al. A histological and functional description of the tissue causing chronic postthrombotic venous obstruction. Thromb Res 2015;135:882–7.
Crossref| PubMed - Saha P, Black S, Breen K, et al. Contemporary management of acute and chronic deep venous thrombosis. Br Med Bull 2016;117:107–20.
Crossref| PubMed - de Wolf MAF, de Graaf R, Kurstjens RLM, et al. Short-term clinical experience with a dedicated venous nitinol stent: initial results with the sinus-venous stent. Eur J Vasc Endovasc Surg 2015;50:518–26.
Crossref| PubMed - Black S, Gwozdz A, Karunanithy N, et al. Two year outcome after chronic iliac vein occlusion recanalisation using the Vici Venous Stent®. Eur J Vasc Endovasc Surg 2018;56:710–18.
Crossref| PubMed - Matsuda A, Yamada N, Ogihara Y, et al. Early and long-term outcomes of venous stent implantation for iliac venous stenosis after catheter-directed thrombolysis for acute deep vein thrombosis. Circ J 2014;78:1234–9.
Crossref| PubMed - Roberts LN, Patel RK, Goss DE, et al. Relationship between development of post-thrombotic syndrome and serial ultrasound, D-dimer, and factor VIII activity after a first deep venous thrombosis. J Vasc Surg Venous Lymphat Disord 2016;4:28–35.
Crossref| PubMed - Siudut J, Grela M, Wypasek E, et al. Reduced plasma fibrin clot permeability and susceptibility to lysis are associated with increased risk of postthrombotic syndrome. J Thromb Haemost 2016;14:784–93.
Crossref| PubMed
